Genetic Basis of Vegetable Grafting: Molecular Communication Between Rootstock and Scion
Vegetable grafting is widely used in modern horticulture to enhance crop productivity, improve resistance to diseases, and increase tolerance to environmental stresses. In this technique, the shoot portion of one plant, known as the scion, is joined with the root system of another plant called the rootstock. Once the two parts are grafted together, they undergo a healing process and eventually grow as a single plant. This approach is commonly applied in vegetable crops such as tomato, cucumber, watermelon, eggplant, and pepper to improve plant vigor, yield stability, and stress adaptability [1].
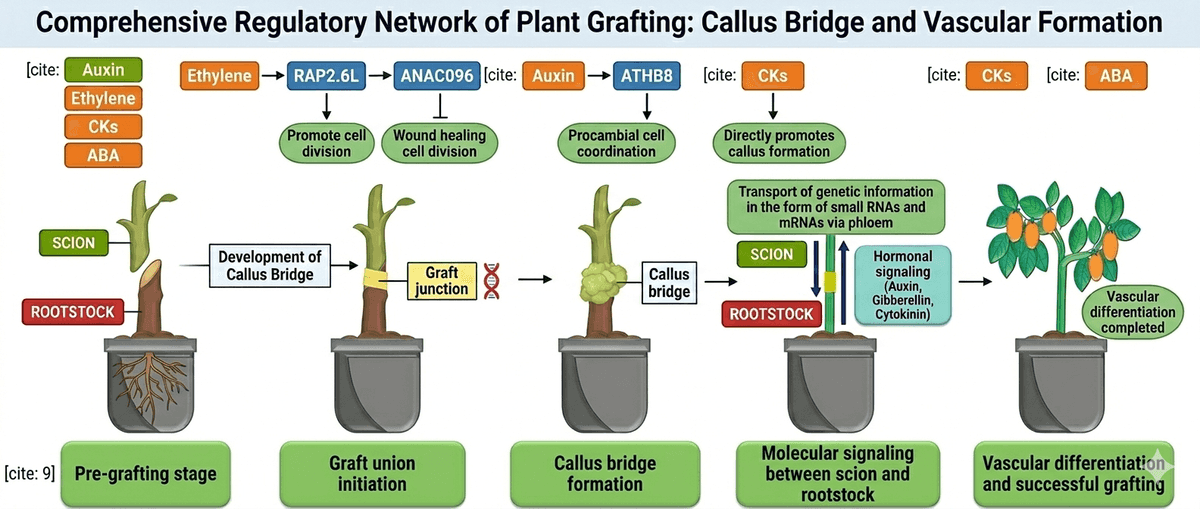
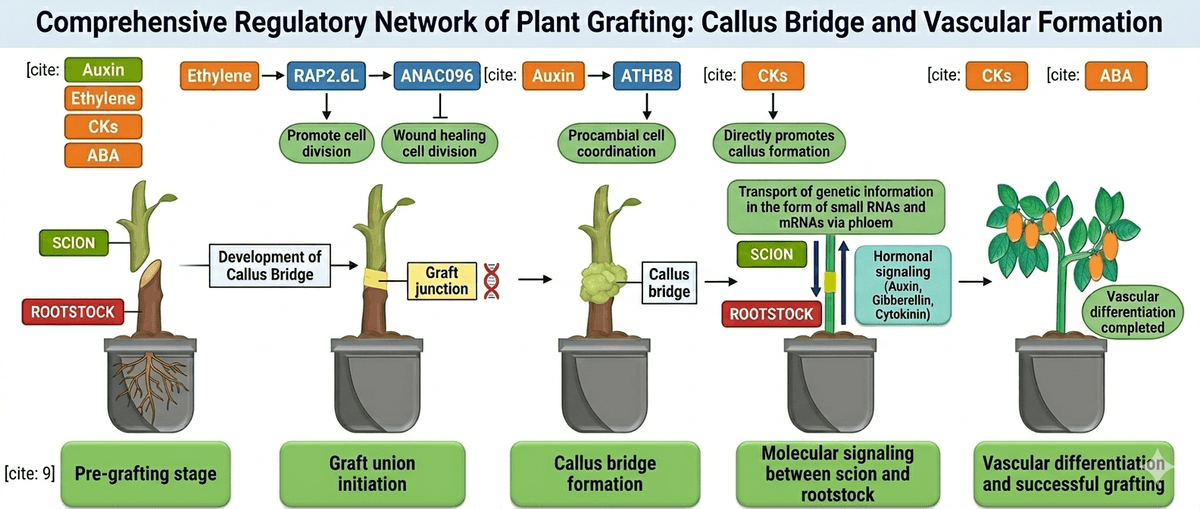
Although grafting has been practiced for centuries, recent studies show that its success is strongly influenced by genetic interactions between the rootstock and scion. After grafting, plants activate a series of genes associated with wound healing, tissue adhesion, and cell proliferation. These genetic responses help repair the damaged tissues at the graft junction and stimulate the formation of callus tissue, which bridges the two plant parts. As healing progresses, cells differentiate to regenerate vascular tissues, allowing the reconnection of xylem and phloem and restoring the transport of water, nutrients, and signalling molecules throughout the plant [2].
Research has also revealed that signaling molecules can move between graft partners through the vascular system, influencing gene expression and plant development. Certain RNA molecules produced in one part of the plant can travel through the vascular system and influence gene expression in the other part. This exchange of genetic signals may regulate plant growth, stress responses, and physiological traits. In some cases, grafting can change plant traits by affecting how genes are expressed, even though the DNA itself does not change [1].
Understanding the genetic basis of vegetable grafting provides valuable insights into how plants communicate, regenerate tissues, and adapt to environmental stresses. With advances in genomics and biotechnology, studying these genetic mechanisms will help improve graft compatibility, enhance crop resilience, and support the development of more sustainable vegetable production systems.
Wound Response and Activation of Healing Genes
The first biological event that occurs after grafting is the wound response, which is triggered when the rootstock and scion are cut and joined together. The cutting damages cells at the graft interface, causing the plant to rapidly activate a repair program that initiates tissue regeneration. Within 24 hours after grafting, wound-responsive genes are activated at the graft junction, initiating the healing process.
Several key transcription factors regulate this early genetic response. One of the important regulators is ANAC071, a NAC-domain transcription factor that promotes tissue reunion by activating genes involved in cell proliferation and cell wall remodelling. Another related gene, ANAC096, works together with ANAC071 and contributes to the formation of cambium-like cells, which later participate in vascular regeneration.
The gene ERF115 (Ethylene Response Factor 115) is also activated during the wound response and helps stimulate cell division and regeneration at damaged tissues. Similarly, WIND1 (Wound-Induced Dedifferentiation 1) plays a key role in promoting cell dedifferentiation, allowing mature cells near the wound to revert to a regenerative state so they can participate in healing. At the same time, genes responsible for cell wall modification are activated to facilitate tissue attachment. For example, GH9B3, a β-1,4-glucanase gene, helps remodel cellulose in the cell wall, enabling cells from the scion and rootstock to adhere more effectively. Other genes such as EXPANSIN (EXP) and PECTATE LYASE-LIKE (PLL) modify pectin and other cell wall components, allowing cell expansion and strengthening tissue adhesion.
Together, these wound-induced genes initiate the regeneration program at the graft interface. As these processes continue, cells surrounding the wound begin to proliferate and form new tissues, leading to the next critical stage of graft healing: callus formation at the graft junction.
Callus Formation and Gene Regulation at the Graft Junction
Following the wound response, the next crucial step in graft healing is callus formation at the graft junction. During this stage, cambium and parenchyma cells located near the cut surfaces of the rootstock and scion begin to divide rapidly, producing a mass of undifferentiated cells known as callus tissue. This callus acts as a biological bridge that gradually connects the tissues of the two plant parts and enables communication between them. This stage generally occurs within 2–3 days after grafting.
The formation and proliferation of callus are regulated by several genes and transcription factors. One of the key regulators is ANAC071, a NAC-domain transcription factor that promotes tissue reunion by activating downstream genes such as XTH19 and XTH20, which are involved in cell wall modification and expansion. These processes allow cells from the rootstock and scion to adhere and proliferate at the graft interface [4].
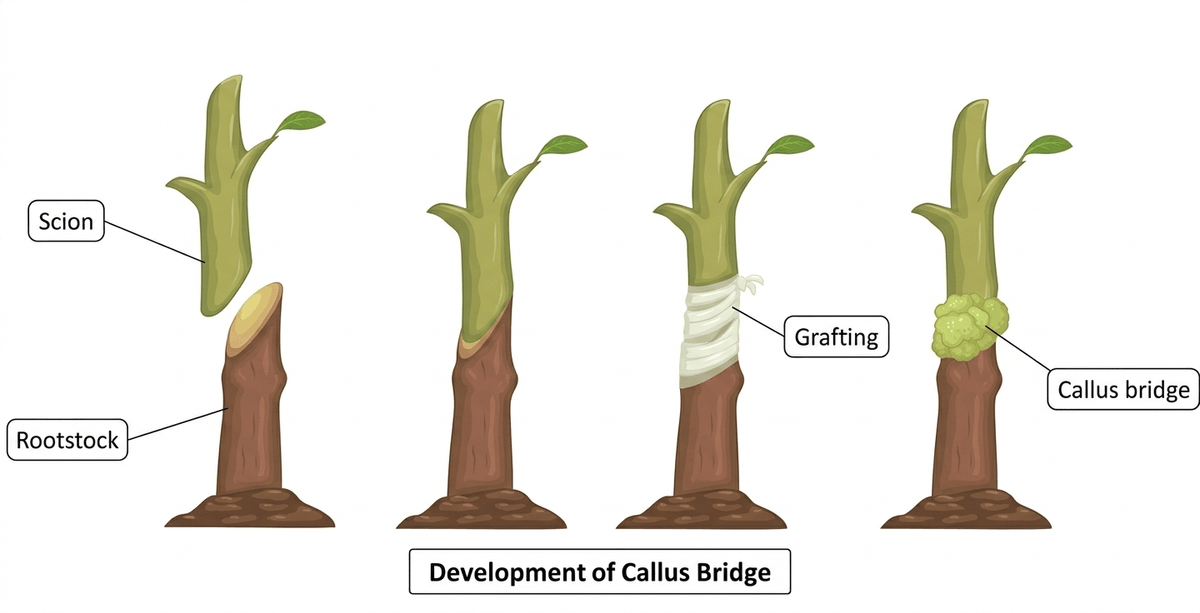
Another transcription factor, RAP2.6L, is expressed mainly on the rootstock side and works together with ANAC071 to regulate cell proliferation and tissue regeneration during graft healing. The activity of these genes is influenced by plant hormones such as auxin, ethylene, and jasmonic acid, which accumulate around the wound site and stimulate cell division. Successful callus formation also requires appropriate environmental conditions. In practical grafting systems, high humidity levels (around 90–95%) are maintained during the first 2–3 days after grafting to prevent scion dehydration and support tissue healing. However, excessive humidity and temperature during the healing stage can promote microbial growth at the graft junction, which may interfere with tissue regeneration and lead to graft failure or plant death [3, 4].
Asymmetrical Gene Expression Between Rootstock and Scion
During the early stages of graft healing, gene expression patterns differ between the rootstock and scion, leading to asymmetrical responses at the graft junction. This asymmetry mainly occurs during the callus proliferation stage, before vascular tissues reconnect. Transcriptomic studies have shown that many differentially expressed genes (DEGs) are activated during graft union formation, particularly those involved in wound response, hormone signaling, oxidative stress response, cell wall synthesis, and vascular tissue development [5, 6].
One of the major causes of this asymmetry is the uneven distribution of sugars between the two graft partners. Sugars tend to accumulate in the scion above the graft union, while the rootstock experiences relative depletion. This imbalance triggers differential expression of sugar-responsive genes such as ApL3, DIN6, GDH1, and STP1, which are associated with sugar metabolism and transport. As vascular tissues reconnect, these differences gradually decrease and gene expression becomes more synchronized between the rootstock and scion [6].
Genetic Control of Cambium Formation (Cambialization)
Cambium formation at the graft junction is regulated by several genes that control cell proliferation and tissue regeneration after grafting. One key regulator is ANAC071, a NAC-domain transcription factor induced by auxin accumulation above the incision through ARF6 and ARF8. ANAC071, together with its homolog ANAC096, promotes the proliferation of cambium-like cells. Another important gene is WIND1 (WOUND-INDUCED DEDIFFERENTIATION 1), which regulates callus formation by activating ESR1 and downstream genes such as PLETHORA3/5/7 and WOX13, stimulating regenerative cell division at the graft interface [7].
Additionally, PAT1 promotes tissue attachment during graft union formation, while AT2-MMP helps regulate the later stages of healing by controlling cell proliferation at the incision site.
Regulation of Vascular Tissue Development
After grafting, the development of functional vascular tissues such as xylem and phloem is essential for reconnecting the rootstock and scion. This process is regulated by several genes and transcription factors involved in vascular differentiation.
Auxin signalling plays a key role in this stage. The transcription factor MONOPTEROS (MP), also known as AUXIN RESPONSE FACTOR 5 (ARF5), regulates vascular development by activating genes required for procambial cell formation. MP directly activates the ATHB8 gene, which is necessary for pre-procambial cell specification and the establishment of vascular tissue identity. In addition, AHP6 (Arabidopsis Histidine Phospho transfer Protein 6) regulates xylem differentiation during vascular regeneration. For phloem development, the BREVIS RADIX (BRX) gene controls the differentiation of protophloem tissues, ensuring proper phloem formation at the graft junction.
Through these coordinated genetic mechanisms, newly formed cambial cells differentiate into functional xylem and phloem, enabling the restoration of water, nutrient, and signal transport between the rootstock and scion. Once vascular tissues reconnect, complex regulatory networks involving plant hormones and transcriptomic changes further coordinate graft union development [8].
Hormonal and Transcriptomic Regulation During Graft Union Formation
During graft union formation, hormonal signaling and transcriptomic regulation coordinate the healing process between the scion and rootstock. Plant hormones such as auxin (IAA), jasmonic acid (JA), ethylene, cytokinins, and abscisic acid (ABA) play key regulatory roles in activating genes involved in tissue regeneration and adventitious root formation. In grafted watermelon–pumpkin systems, increased IAA levels in the rootstock were associated with improved adventitious root development, while relatively lower ABA and JA levels supported root initiation and growth after grafting.
Transcriptomic analyses reveal extensive differential gene expression during graft healing. Many genes involved in hormone signaling pathways become activated, including auxin signaling components such as AUX1 transporters, TIR1 receptors, Aux/IAA repressors, ARF transcription factors, GH3 genes, and SAUR genes, indicating strong auxin-mediated regulation during graft union development. In addition, transcription factors such as bHLH, ERF, MYB, and NAC are significantly expressed and regulate hormone-responsive gene networks that control cell division and tissue regeneration [9].
Another important mechanism involves cell wall remodeling at the graft interface. Transcriptomic studies in oriental melon–squash graft systems identified CmGH9B3, a β-1,4-glucanase gene, as a key regulator of graft healing. This enzyme participates in cell wall modification and reconstruction, allowing cells from the scion and rootstock to adhere and form a continuous tissue connection during graft union formation [4, 9].
Sugar Signaling and Epigenetic Regulation in Grafted Plants
Sugar signaling plays an important role in regulating gene expression during graft union formation. Differences in sugar accumulation between the scion and rootstock can influence the expression of sugar-responsive genes and affect metabolic and transcriptional processes in grafted plants. Changes in carbohydrate metabolism and sugar distribution are closely linked with transcriptional alterations that occur after grafting, which can influence plant development and stress responses [10].
Grafting also involves epigenetic regulation, where gene expression is modified without altering the DNA sequence. Epigenetic changes mainly occur through mechanisms such as DNA methylation, histone modification, and non-coding RNAs, which regulate chromatin structure and gene activity in plants. These epigenetic processes are involved in controlling plant growth, development, and responses to environmental conditions [11].
Mobile signaling molecules can move between the rootstock and scion through the vascular system and trigger epigenetic changes in recipient tissues. In particular, small RNAs (miRNAs and siRNAs) can travel across the graft junction through the phloem and regulate gene expression by inducing post-transcriptional gene silencing or RNA-directed DNA methylation. These processes modify the epigenome of recipient tissues and may influence plant architecture, stress tolerance, and other physiological traits [12]. Through these mechanisms, sugar signaling and epigenetic regulation contribute to the coordination of gene expression between rootstock and scion, influencing the physiological performance and adaptability of grafted plants.
Importance and Future Perspectives of Genetic Studies in Vegetable Grafting
Understanding the genetic basis of vegetable grafting is important because it explains how rootstock and scion successfully heal and function as a single plant. Genetic regulation controls key processes such as wound response, callus formation, cambium development, and vascular reconnection, which ultimately determine graft compatibility and union success. Knowledge of these mechanisms helps identify important genes and regulatory pathways that influence tissue regeneration and communication between graft partners.
This understanding also provides practical benefits for agriculture by enabling the selection of improved rootstocks that enhance disease resistance, stress tolerance, nutrient uptake, and overall plant vigor. As environmental stresses and climate change increasingly affect vegetable production, genetically informed grafting strategies can help develop more resilient cropping systems.
In the future, advances in genomics, transcriptomics, and gene-editing technologies may further reveal the molecular networks involved in graft healing and support the development of more efficient and sustainable vegetable grafting practices.
Genes Involved in Vegetable Graft Healing and Union Formation
| S. No | Gene Category | Genes |
|---|---|---|
| 1. | Auxin Response Factors | ARF5(MONOPTEROS/MP), ARF6, ARF8 |
| 2. | Auxin Signaling Genes | Aux/IAA, GH3, SAUR |
| 3. | Auxin Signaling Proteins | AUX1, TIR1 |
| 4. | Cell Proliferation Regulators | AT2-MMP, ESR1 |
| 5. | Cell wall modification enzymes | CmGH9B3, EXP, GH9B3, PLL, XTH19, XTH20 |
| 6. | Metabolic regulation genes | GDH1 |
| 7. | NAC transcription factors | ANAC071, ANAC096 |
| 8. | Phloem development gene | BRX |
| 9. | Regeneration regulators | PLETHORA3, PLETHORA5, PLETHORA7, WOX13 |
| 10. | Sugar metabolism genes | ApL3, DIN6 |
| 11. | Sugar transporter | STP1 |
| 12. | Tissue attachment regulator | PAT1 |
| 13. | Transcription factors | RAP2.6L |
| 14. | Transcription factor families | bHLH, ERF, MYB, NAC |
| 15. | Vascular development genes | AHP6, ATHB8 |
| 16. | Wound-induced regulator | WIND1 |
| 17. | ERF transcription factor | ERF115 |
Key Takeaways
- Vegetable grafting is a powerful horticultural technique that improves crop yield, disease resistance, and tolerance to environmental stresses by combining the strengths of rootstock and scion.
- Graft healing begins with a wound response, where plants activate specific genes within about 24 hours to start tissue repair and regeneration.
- Callus formation acts as the first physical bridge between rootstock and scion, allowing the two plant parts to gradually connect and communicate.
- Early graft healing shows asymmetrical gene expression, mainly due to differences in sugar distribution and metabolic activity between the two graft partners.
- Cambium formation and vascular reconnection are essential steps, enabling the development of new xylem and phloem that restore water, nutrients, and signal transport.
- Plant hormones such as auxin, ethylene, jasmonic acid, and cytokinin coordinate graft healing by regulating genes involved in cell division and tissue regeneration.
- Sugar signaling and mobile RNAs help rootstock and scion communicate, influencing gene expression and physiological responses across the graft junction.
- Epigenetic processes like DNA methylation can influence plant traits, sometimes leading to changes in growth, stress tolerance, or fruit quality.
- Advances in genomics and biotechnology are improving our understanding of graft healing, which may help develop better graft compatibility and more resilient vegetable crops in the future.
References
[1] Majhi PK, Bhoi TK, Sahoo KC, Mishra N, Tudu S, Das S, Ray M, Singh S, Samal I, Behera PP, Saini V. Understanding the genetics and genomics of vegetable grafting to ensure yield stability. In Smart plant breeding for vegetable crops in post-genomics era 2023 Jan 1 (pp. 69-98). Singapore: Springer Nature Singapore.
[2] Feng M, Augstein F, Kareem A, Melnyk CW. Plant grafting: Molecular mechanisms and applications. Molecular Plant. 2024 Jan 1;17(1):75-91.
[3] Lixian Wang, Yangmei Liao, Jiming Liu, Tianyun Zhao, Liming Jia, Zhong Chen, Advances in understanding the graft healing mechanism: a review of factors and regulatory pathways, Horticulture Research, Volume 11, Issue 8, August 2024, uhae175
[4] Nie W, Wen D. Study on the applications and regulatory mechanisms of grafting on vegetables. Plants. 2023 Jul 30;12(15):2822.
[5] Shivran M, Sharma N, Dubey AK, Singh SK, Sharma N, Sharma RM, Singh N, Singh R. Scion–rootstock relationship: Molecular mechanism and quality fruit production. Agriculture. 2022 Nov 28;12(12):2036.
[6] Huang W, Wang S, Mao C, Xiang L, Zhang X, Jiang F, Cheng Y, Li T. Integrative Analyses of Metabolome and Transcriptome Reveal Scion–Stock Asymmetry Reduction and Shift of Sugar Metabolism During Graft Junction Formation in Malus Domestica (‘Hanfu’) Homograft. International Journal of Molecular Sciences. 2025 May 30;26(11):5290.
[7] Xiong M, Zhang T, Qian X, Kadeer A, Kurotani KI, Li L, Liu C, Wu X, Bie Z, Notaguchi M, Huang Y. Xyloglucan endotransglucosylase/hydrolase family genes are required for the plant graft union formation through callus proliferation. Plant Physiology. 2026 Jan 29:kiag030.
[8] Ahammed, G. J., & Zhou, J. (2025). Introduction to vegetable crop growth regulation and quality improvement. In G. J. Ahammed & J. Zhou (Eds.), Growth regulation and quality improvement of vegetable crops: Physiological and molecular features. Springer Nature.
[9] Song C, Huang Y, Wu C, Tian B, Shi X, Mi G, Jing Y, Tang Y, Wang Z, Niu L, Wang T. Scion organ removal alters hormone levels and gene expression associated with adventitious root development in grafted watermelon seedlings. Plant Signaling & Behavior. 2025 Dec 31;20(1):2556300.
[10] Jeynes-Cupper K, Catoni M. Long distance signalling and epigenetic changes in crop grafting. Frontiers in Plant Science. 2023 Mar 20;14:1121704.
[11] Jin Q, Chachar M, Ahmed N, Zhang P, Chachar Z, Geng Y, Guo D, Chachar S. Harnessing epigenetics through grafting: revolutionizing horticultural crop production. Horticulturae. 2023 Jun 6;9(6):672.
[12] Ruan M, Huang X, Wang R, Cheng Y, Zhou G, Ye Q, Yao Z, Wan H, Li Z, Liu C, Zhang C. Eggplant Rootstock Grafting Enhances Tomato Fruit Sweetness and Nutritional Value via Metabolic Reprogramming. Food Science & Nutrition. 2026 Jan;14(1):e71431.
Published on: March 4, 2026